Your Location:Home >Products >API >120202-66-6

Product Details
|
Description |
(S)-(+)-Clopidogrel is the functional enantiomer of clopidogrel and a prodrug whose thiol metabolite antagonizes purine binding to the platelet purinergic receptor P2Y12 (Ki = 316 nM in human platelets). It is metabolized by the cytochrome P450 (CYP) isoform CYP2C19 in rat liver and inhibits ADP-induced platelet aggregation ex vivo. Formulations containing (S)-(+)-clopidogrel have been used in combination with aspirin to prevent vascular ischemic events in patients with acute coronary syndromes. |
|
Chemical Properties |
Off-White Solid |
|
Originator |
Iscover,Bristol-Myers |
|
Uses |
Clopidogrel Bisulfate is an oral, thienopyridine class antiplatelet agent used to inhibit blood clots in coronary artery disease, peripheral vascular disease, and cerebrovascular disease. |
|
Manufacturing Process |
Levo-rotatory ammonium camphor-10-sulfonate is dissolved in a minimum of water and applied to the column of Amberlite IRN-77 resin. Elution is carried out with water. The eluted fractions containing the levo-rotatory camphor-10- sulfonic acid are lyophilized, melting point 198°C. 32 g (0.0994 mole) of racemic methyl-α-5-(4,5,6,7-tetrahydro-thieno(3,2- c)pyridyl)(2-chlorophenyl)-acetate are dissolved in 150 ml of acetone. 9.95 g (0.0397 mole) of levo-rotatory camphor-10-sulfonic acid monohydrate are added. The clear solution is left to stand at room temperature. After 48 hours the reaction mixture is concentrated to 50 ml and left to stand at room temperature for 24 hours. The obtained camphor-10-sulfonic acid salt of methyl-α-5-(4,5,6,7-tetrahydro-thieno(3,2-c)pyridyl)(2-chlorophenyl)-acetate (SR 25990) are filtered off, washed with acetone and dried (yield: 55% on the basis of the starting racemate), melting point 165°C, [α]D20=+24.67 (c=1.58 g/100 ml; methanol). The crystals obtained above are redissolved in the minimum of boiling acetone (50 ml). The crystals obtained after cooling are filtered off, washed with acetone and dried (yield: 88%), m.p. 165°C, [α]D20=+24.75 (c=1.68 g/100 ml; methanol). 12 g (0.022 mole) of the pure camphor-10-sulfonic acid salt of methyl-α-5- (4,5,6,7-tetrahydro-thieno(3,2-c)pyridyl)(2-chlorophenyl)-acetate are dissolved in a minimum of water. After cooling to 5°C, the aqueous solution obtained is made alkaline with a saturated aqueous solution of sodium hydrogen carbonate. The alkaline aqueous phase is extracted with dichloromethane. The organic extracts are dried over anhydrous sodium sulfate. On evaporation of the solvent a colorless oil of dextro-rotatory methyl-α-5-(4,5,6,7-tetrahydro-thieno(3,2-c)pyridyl)(2-chlorophenyl)-acetate is obtained (quantitative yield). Oil, [α]D20=+51.52 (c=1.61 g/100 ml; methanol). 800 ml of a saturated aqueous solution of sodium bicarbonate are added to a suspension of 200 g of SR 25990 in 800 ml of dichloromethane. After vigorous shaking, the organic phase is separated, dried over sodium sulfate and the solvent is removed under reduced pressure. The residue is dissolved in 500 ml of ice-cold acetone and 20.7 ml of concentrated sulfuric acid (93.64%) areadded drop-wise. The precipitate formed is isolated by filtration and washed with 1 L of acetone, then dried in a vacuum oven at 50°C. 139 g of pure white crystals of hydrogen sulfate of dextro-rotatory methyl-α-5-(4,5,6,7- tetrahydro-thieno(3,2-c)pyridyl)(2-chlorophenyl)-acetate (SR 25990 C) are thus obtained, m.p. 184°C, [α]D20=+55.10 (c=1.891 g/100 ml; methanol). |
|
Brand name |
Plavix (Sanofi Aventis). |
|
Therapeutic Function |
Platelet aggregation inhibitor |
|
General Description |
Clopidogrel bisulfate (CLP) is an antiplatelet drug, which belongs to the thienopyridine class of drug. |
|
Biochem/physiol Actions |
(S)-(+)-Clopidogrel hydrogen sulfate is an antithrombotic antiplatelet agent. It specifically and irreversibly inhibits the Purinoceptor P2Y12 subtype which inhibits ADP-induced platelet aggregation. (S)-(+)-Clopidogrel hydrogen sulfate is the active isomer. |
|
Veterinary Drugs and Treatments |
Clopidogrel, a platelet aggregation inhibitor, may be useful for preventing thrombi in susceptible cats. It may also improve pelvic limb circulation in cats after a cardiogenic embolic event via a vasomodulating effect secondary to inhibition of serotonin release from platelets. Research is ongoing. |
InChI:InChI=1/C15H14ClNO2S.H2O4S/c16-12-4-2-1-3-11(12)14(15(18)19)17-7-5-13-10(9-17)6-8-20-13;1-5(2,3)4/h1-4,6,8,14H,5,7,9H2,(H,18,19);(H2,1,2,3,4)/t14-;/m0./s1
In this work primarily one-pot synthetic...
Copper (II) bromide catalyzed one pot α-...
The invention relates to a preparation m...
The present invention relates to a prepa...
The invention discloses a preparation me...
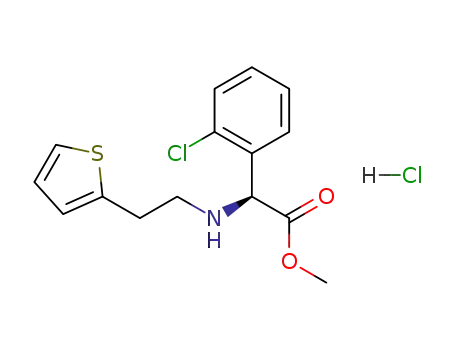
(S)-(+)-α-(2-thienylethylamino)-α-(2-chlorophenyl)acetic acid methyl ester hydrochloride

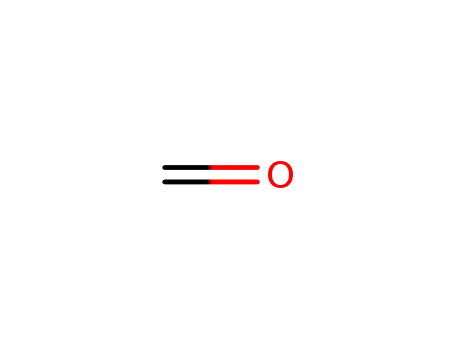
formaldehyd

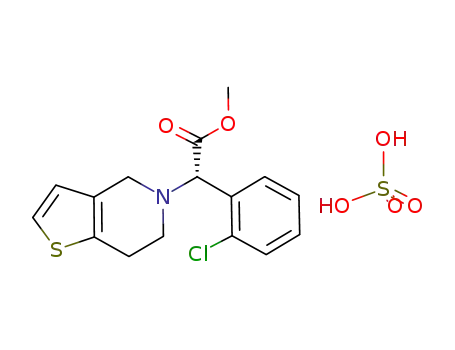
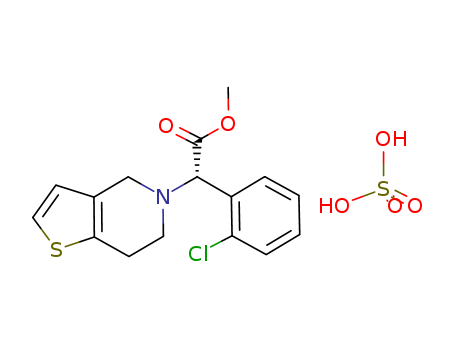
(S)-(+)-clopidogrel bisulfate
| Conditions | Yield |
|---|---|
|
(S)-(+)-α-(2-thienylethylamino)-α-(2-chlorophenyl)acetic acid methyl ester hydrochloride; formaldehyd; In methanol; at 43 ℃; for 5h;
With sulfuric acid; at 38 ℃; for 4h; Temperature;
|
100% |
|
(S)-(+)-α-(2-thienylethylamino)-α-(2-chlorophenyl)acetic acid methyl ester hydrochloride; formaldehyd; hydrogenchloride; In water; at 80 - 85 ℃; for 1h;
With sulfuric acid; In methanol; acetone; at 0 - 30 ℃; for 15h;
|
99.6 % ee |
|
(S)-(+)-α-(2-thienylethylamino)-α-(2-chlorophenyl)acetic acid methyl ester hydrochloride; formaldehyd; at 25 - 55 ℃;
With sulfuric acid; In acetic acid butyl ester; at -10 - 0 ℃;
|
|
|
(S)-(+)-α-(2-thienylethylamino)-α-(2-chlorophenyl)acetic acid methyl ester hydrochloride; formaldehyd; In cyclohexane; water; at 40 ℃; for 6h; Large scale;
With sulfuric acid; In methanol; acetone; at -5 ℃; for 8h; Solvent; Temperature; Concentration; Large scale;
|
72 kg |
|
In water; at 36 - 44 ℃;
|
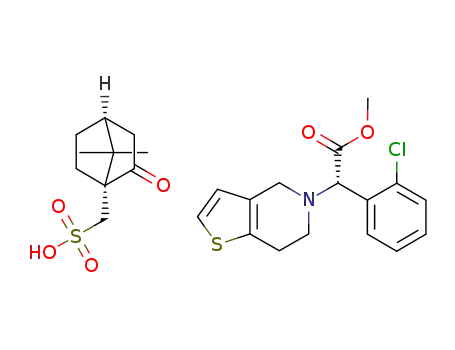
clopidogrel (1R)-(-)-camphor-10-sulfonate


(S)-(+)-clopidogrel bisulfate
| Conditions | Yield |
|---|---|
|
With sulfuric acid; In ethyl acetate; at 10 ℃; for 4h; Solvent;
|
85% |
|
With sulfuric acid; In acetic acid butyl ester; acetic acid;
|
170.5 g |
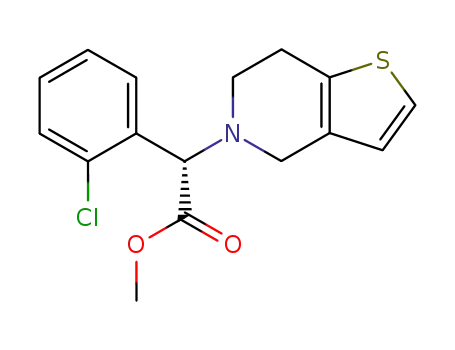
(S)-(+)-clopidogrel
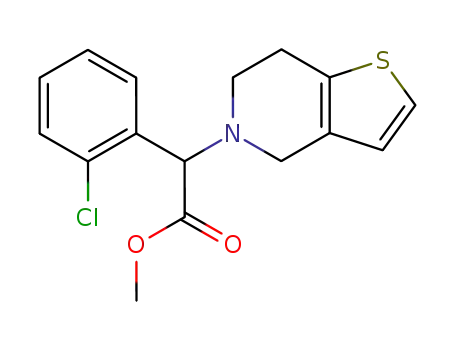
clopidogrel
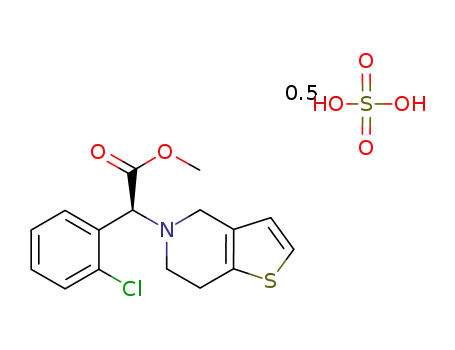
clopidogrel hydrogen sulfate
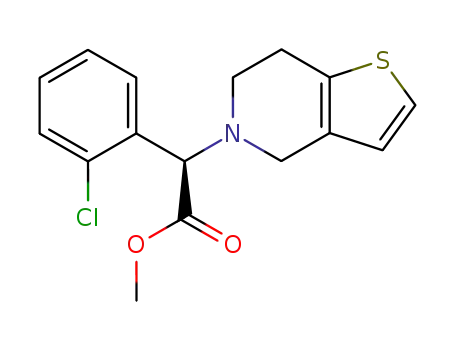
methyl (R)-2-(2-chlorophenyl)-2-(4,5,6,7-tetrahydrothieno[3,2-c]pyridin-5-yl)acetate
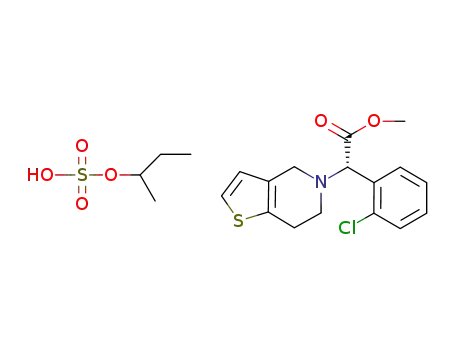
clopidogrel-sec-butyl sulfate

clopidogrel hydrogen sulfate

(S)-(+)-clopidogrel
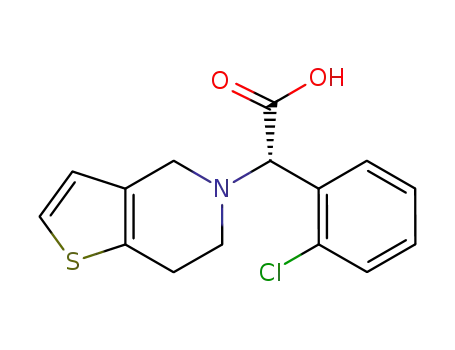
SR 26334
CAS:97-08-5
CAS:13360-57-1
CAS:128794-94-5
CAS:78415-72-2